Imagine holding a warm cup of coffee on a chilly morning or feeling a cool breeze on a hot day. These everyday experiences are governed by the fundamental principles of heat transfer in physics, specifically conduction and convection. These phenomena play a crucial role in various natural processes and technological applications, from the workings of our planet’s atmosphere to the design of heating and cooling systems.

✅ AI Essay Writer ✅ AI Detector ✅ Plagchecker ✅ Paraphraser
✅ Summarizer ✅ Citation Generator
What is Conduction?
Conduction is a mode of heat transfer that occurs through direct molecular collision within a material without any actual movement of the material as a whole. It is the process by which thermal energy is transferred from a region of higher temperature to a region of lower temperature within a solid or between solids in physical contact. The rate of heat transfer by conduction is governed by Fourier’s law, which states that the heat flow rate (q) through a material is proportional to the temperature gradient (ΔT) and the cross-sectional area (A), and inversely proportional to the thickness (d) of the material:
where k is the thermal conductivity of the material, a measure of its ability to conduct heat. Materials with high thermal conductivity, such as metals, are good conductors of heat, while those with low thermal conductivity, such as wood or foam, are considered insulators.
Conduction plays a vital role in our daily lives. It is the phenomena that governs the simple act of warming our hands by holding a hot cup, as well as the complex functioning of electronic devices, where it is responsible for dissipating heat to prevent overheating.
What is Convection?
Convection is a mode of heat transfer that involves the movement of a fluid (liquid or gas) due to differences in temperature and density within the fluid. When a fluid is heated, it expands, becomes less dense, and rises, while cooler, denser fluid sinks. This movement creates a convective current, which is responsible for the transfer of thermal energy throughout the fluid.
The rate of heat transfer by convection can be expressed by Newton’s law of cooling:
where q is the heat transfer rate, h is the convective heat transfer coefficient, A is the surface area through which heat is transferred, and ΔT is the temperature difference between the fluid and the surface.
Convection can be commonly observed in everyday life. For example, you can see it in the process of boiling water, the circulation of air in the atmosphere, and the cooling of a hot cup of tea. It is also a critical process in various engineering applications, including the design of heating, ventilation, and air conditioning (HVAC) systems, and the cooling of electronic components.
Conduction vs. Convection: Key Differences
At a first glance, you may think that conduction and convection sound as similar notions. However, when you dig into these notions deeper (under the surface definitions), you will see that they couldn’t be any more different.
Firstly, the medium in which these processes occur is quite different. Conduction can happen in solids, liquids, and gases, but it’s most effective in solids. Picture a metal spoon heating up in a pot of hot soup. The heat travels through the spoon by conduction, from the hot end submerged in the soup to the cooler end you’re holding. In contrast, convection is exclusive to fluids, which include both liquids and gases. An example of convection is the warm air rising from a heater, circulating throughout the room and transferring heat as it moves.
Now, let’s talk about the mechanism behind these processes. Conduction is all about the transfer of heat through the vibrations and collisions of particles within a material. There’s no large-scale movement of the material itself; it’s a kind of “whisper down the lane” at the molecular level. Convection, however, involves the actual movement of fluid particles. When water is heated in a pot, the warmer, less dense water rises while cooler, denser water sinks, creating a convective current that distributes heat throughout the pot.
The direction of heat flow also sets these two processes apart. In conduction, heat travels in a straight line from areas of higher temperature to areas of lower temperature. Convection is a bit more adventurous, as the direction of heat transfer depends on the movement of the fluid, which can be upward, downward, or sideways, depending on the specific situation.
Finally, let’s consider the speed of these processes. Conduction is generally a slower process, as it relies on the gradual transfer of energy from particle to particle. Convection, on the other hand, can move heat much more quickly over larger distances, thanks to the fluid motion that carries thermal energy along with it.
Here’s a quick comparison table so you can better memorize all the mentioned differences
| Conduction | Convection | |
|---|---|---|
| Solids, liquids, gases | Medium | Fluids (liquids and gases) |
| Molecular collisions | Mechanism | Bulk fluid movement |
| Direction of decreasing temperature | Direction | Influenced by fluid movement |
| Slower | Speed | Faster |
So, while both conduction and convection play crucial roles in heat transfer, they have their own unique characteristics and applications. Understanding these differences helps us appreciate the complex and dynamic ways in which heat moves around us.
Conclusion
As you can see, the principles of conduction and convection are essential for understanding various ways in which heat is transferred in our surroundings. From the warmth of a cozy blanket to the cooling breeze of a fan, these phenomena are integral to our daily experiences and the functioning of countless technological systems.


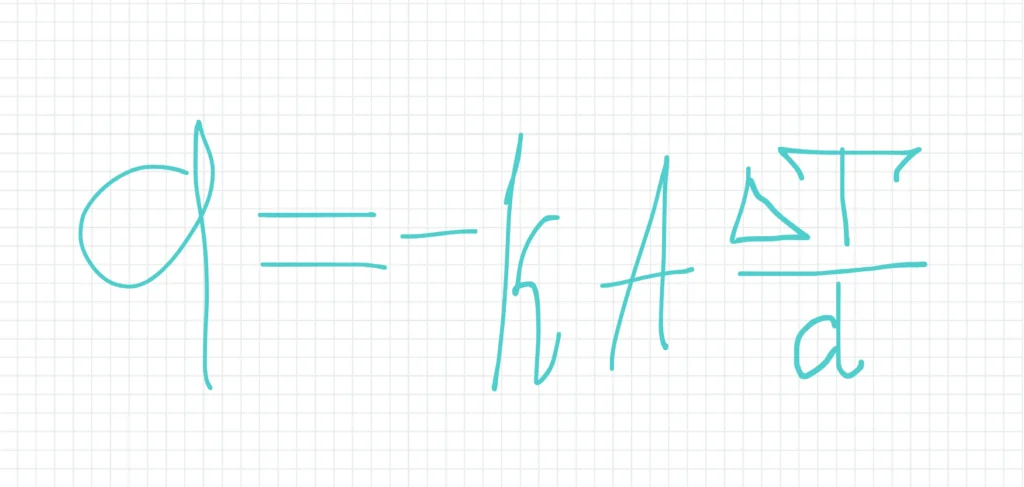
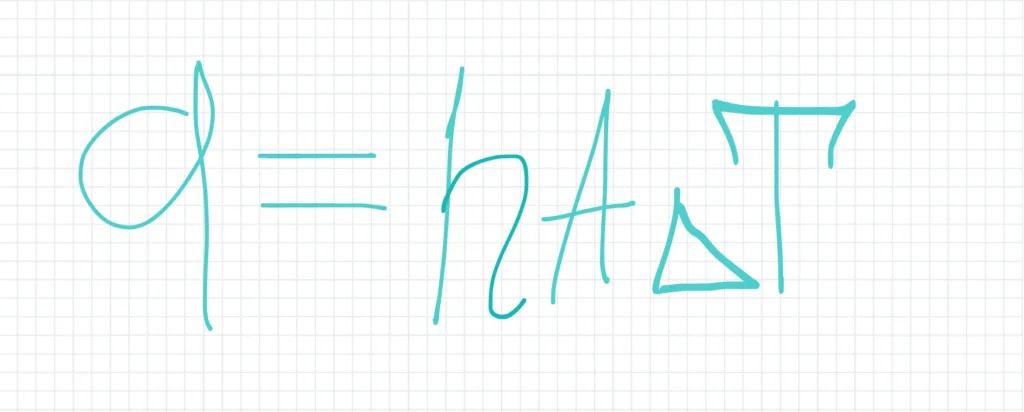


Comments (0)
Welcome to A*Help comments!
We’re all about debate and discussion at A*Help.
We value the diverse opinions of users, so you may find points of view that you don’t agree with. And that’s cool. However, there are certain things we’re not OK with: attempts to manipulate our data in any way, for example, or the posting of discriminative, offensive, hateful, or disparaging material.