K has many different meanings in physics. From its role in defining the stiffness of springs to its significance in electrostatic interactions, the term ‘K’ is multifaceted. In this article, we will discuss some of the most common meanings of K in physics, diving deep into Hooke’s law and Coulomb’s law to understand the underlying principles and their implications.

✅ AI Essay Writer ✅ AI Detector ✅ Plagchecker ✅ Paraphraser
✅ Summarizer ✅ Citation Generator
K in Hooke’s Law
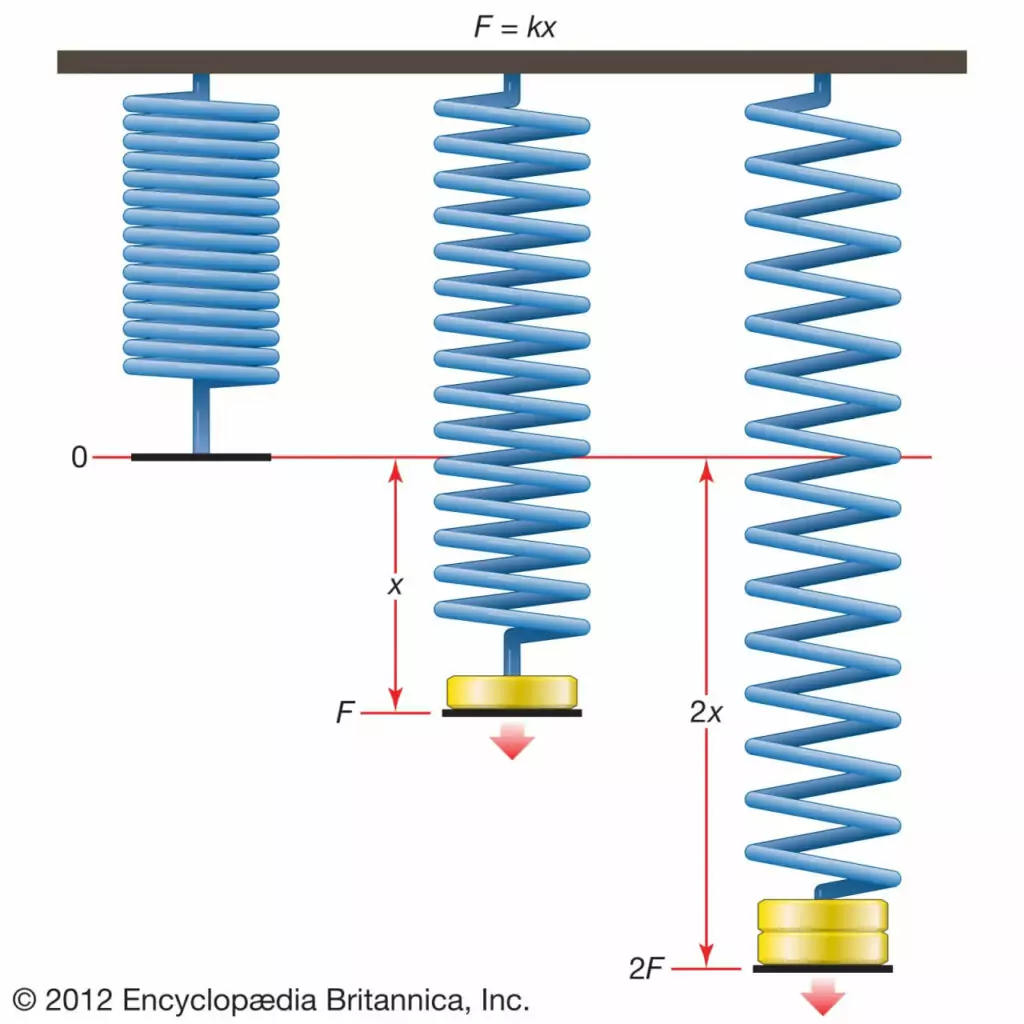
Hooke’s law defines the relationship between the force applied to a spring and its subsequent deformation.
Represented by the formula F=−kx, where:
- F is the force in Newtons (N)
- k is the spring constant (N/m), denoting stiffness
- x is the displacement or how much the spring is stretched or compressed in meters (m)
This law is foundational in understanding the behavior of springs and many materials that demonstrate elastic properties.
Visualizing the Spring Constant
Imagine a toy spring. When you pull on it gently, it elongates slightly. If you apply double the force, it stretches twice as much. This linear relationship between force and displacement signifies the proportionality constant, ‘k’. If ‘k’ is large, it means that even a small stretch requires a lot of force. Thus, the spring is considered stiff. Conversely, a small ‘k’ value means the spring is soft and easy to deform.
However, it’s essential to understand that Hooke’s law isn’t universally accurate. Beyond a certain point, materials can deform permanently, and their response to force is no longer linear. For example, if you stretch a rubber band too much, it doesn’t return to its original length, and the law doesn’t apply.
K in Electrostatic Force (Coulomb’s Law)
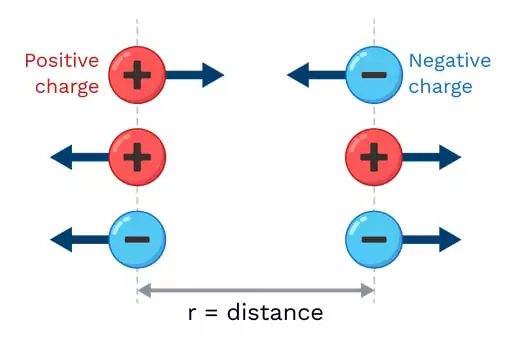
The realm of electrostatics deals with the interactions between static electric charges. Coulomb’s law is a fundamental principle in this area, explaining the forces between charged bodies.
Understanding the Dielectric Constant
Coulomb’s law states that the force of attraction or repulsion between two charges is directly proportional to their product and inversely proportional to the distance squared between them. When charges are in a medium other than a vacuum, their interaction is affected by the medium’s dielectric properties. The dielectric constant ‘k’ in Coulomb’s law represents this influence.
Visually, think of two charges in open air being drawn together. Now, imagine placing a sheet of plastic (a dielectric material) between them. Their attraction becomes less intense because of the plastic’s dielectric properties. The dielectric constant quantifies this change, with a value of 1 for vacuum (no influence) and greater than 1 for other materials.
Defining Coulomb’s Constant
K in Coulomb’s law, often referred to as Coulomb’s constant, electrostatic constant, or electrical force constant, signifies the strength of the electrostatic interaction. Its mathematical representation is:
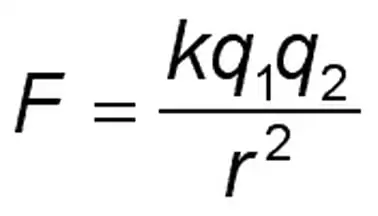
Where:
- F is the electrostatic force
- K is the Coulomb’s constant, with a value of 8.99×109 N.m2/C2
- 1q1 and 2q2 are the charges
- r is the distance between them
This constant is foundational in determining the magnitude and direction of the force between charged entities.
Visualizing Coulomb’s Constant
Imagine two magnets. Their force of attraction depends on their size, distance, and the medium between them. Similarly, the force between two electric charges depends on their magnitude, distance apart, and the properties of the intervening medium. The Coulomb’s constant provides a baseline to calculate this force, considering the effects of the medium.
In conclusion, ‘K’ is a versatile term in physics, playing pivotal roles in varied domains, from mechanical systems like springs to the intricate world of electrostatic interactions. Understanding its significance and application is vital for grasping many of the laws and principles that govern our universe. Additionally, if you’re studying physics, you may come across the elastic potential energy formula, which involves the spring constant (K) and the displacement of the spring from its equilibrium position.





Comments (0)
Welcome to A*Help comments!
We’re all about debate and discussion at A*Help.
We value the diverse opinions of users, so you may find points of view that you don’t agree with. And that’s cool. However, there are certain things we’re not OK with: attempts to manipulate our data in any way, for example, or the posting of discriminative, offensive, hateful, or disparaging material.